Chlorine is an atom with an atomic number of 17 and atomic weight of 35, meaning that it has 17 electrons and protons and 18 neutrons as shown in the diagram on the left (Gagnon, 2016). It is situated on the right of the periodic table 2nd last group. This makes chlorine a halogen along with fluorine and bromine. Being a halogen means that chlorine is highly reactive and poisonous. Chlorine is on the 3rd period and 7th group meaning it has 3 shells and 7 electrons on the third shell (Gagnon, 2016). This also demonstrates how reactive chlorine is due to the fact that it is missing 1 electron and therefore needs to bond to have a complete outer shell. Since it bonds and can combine directly with nearly every element, chlorine is never found roaming around in nature. Chlorine is commonly used as an antiseptic used to make drinking water safe and swimming pools clean (Gagnon, 2016). One of the most familiar chlorine compounds are sodium chloride/ table salt which will be discussed further on in the website.
Bonding
Like many other elements, chlorine isn’t fixed to one type of bonding. In fact it undergoes 2 types; ionic and covalent. This is because “chlorine” doesn’t care how it gets a full outer shell, it just cares that it does. There are many different elements that are suitable to perform covalent bond with chlorine and some can bond ionically with chlorine. Below are two examples that demonstrate how chlorine undergoes both covalent and ionic bonding and why.
|
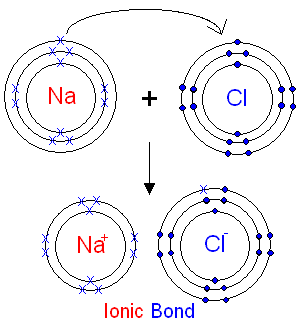
Ionic bonding
During ionic bonding, one atom gives/donates an electron to another atom which receives the donated electrons. Ionic bonding is performed when one atom needs to lose X amount of electrons to have a full outer shell and another atom needs that same X amount of electrons to be full. Then the two atoms bond ionically and give and receive electrons to become in their stable state (their dream). In this case, you can see ionic bonding between sodium and chlorine. As mentioned before, chlorine has 7 electrons on it’s outer shell and therefore needs one more to have a full outer shell of 8. In contrast, sodium needs to lose one electron to have a full outer shell of 8. Therefore it would make sense for sodium to lose its one electron and donate that to a “needy” atom which in this case is chlorine (Unknown, 2015). Now that the two atoms have donated and received one electron, both have a full outer shell and have ionically bonded. Although now both atoms have a different amount of electrons compared to protons. Sodium now only has 10 negatively charged electrons and 11 positively charged protons, whereas chlorine has 18 negatively charged electrons and have 17 positively charged protons. This means that both atoms are now ions. Sodium is now a cation/positively charged atom and chlorine is an anion/a negatively charged atom (Unknown, 2015). On the provided diagram on the left we can see the process of ionic bonding in a more visual way. |
|
Covalent bonding
When an atom undergoes covalent bonding, electrons are shared between the two atoms so that both elements can achieve a full outer shell. We can see chlorine go through this binding process in the diagram on the right. Both chlorine atoms have 7 electrons on the outer shell and therefore both need one electron to complete their outer shell. Now the two atoms will undergo covalent bonding and will “share” their electron. Chlorine atom X shares its one atom with chlorine atom B and both now have a completed and full outer shell (Clarke, 2014). By sharing the two electrons where the shells touch each chlorine atom can count 8 electrons in its outer shell. Now the two atoms are completed/have a full outer shell and are stable and have now created the Cl2 molecule that is created with 2 chlorine atoms. Because the two atoms are now stable they will not go through anymore covalent bonding (it is very unlikely) because it already has a full shell, is stable and consequently less reactive (Clarke, 2014). Contrasting to ionic bonding, there are no ions. This is because no atom has fully received or fully donated an electron, instead the electrons are shared. The diagram to the right shows these processes. |
Physical properties
The Physical properties of Chlorine are the characteristics that can be observed without changing the composition of the matter/ turning the substance into another substance. We use physical properties to describe and observe the substance. Physical properties are usually those that can be observed using our senses such as color, luster, freezing point, boiling point, melting point, density, hardness and odor. An example of a physical property of chlorine is that in room temperature it is a yellowish-greenish colour (Unknown, 2016). Chlorine is denser than air (about 1 ½ times denser). Additionally chlorine’s boiling point is about -35/-34 degrees celsius, this means that it will turn solid when it reaches -34/-35 degrees celsius (Unknown, 2016). Although this may seem like we are changing the chlorine to liquid to find its boiling point it isn’t. This is because although the state of matter may change, the composition of the substance is completely the same. Chlorine has a suffocating smell and inhalation causes suffocation, constriction of the chest, tightness in the throat etc.
Chemical properties-element chlorine
Chemical properties are the characteristics that determine how it will react with other substances or change from one substance to another. We can only observe chemical properties during a chemical reaction (Leon, 2016). An example of chlorine is that it’s chemical formula is Cl. It is highly toxic, this is a chemical property since we would need to test how toxic it is which would cause some sort of reaction. Chlorine also has immediate solubility in water, and when it is added into water dissociation occurs and the water breaks down chlorine (Unknown, 2016). Chlorine is highly reactive (as mentioned before) and will react with nearly everything in nature which is why you won’t be able to see chlorine in its natural form in nature. Although some reactions can be extremely violent, especially those with hydrocarbons, alcohols etc.
Evaluation
So now that we now what physical and chemical properties chlorine has, what does it mean? Well, for example a physical property of chlorine is that it is a gas, as demonstrated in the particle model the particles in gases move very quickly and have a lot of space between them. This can be a reason why chlorine is so reactive (Banks, 2016). With the added fact that chlorine only has 7 electron on the outer shell. Because it only has 7 it needs 1 more electron to achieve a full outer shell with 8 electrons. Therefore the chlorine atoms are actively searching for other atoms to bond with to achieve a full outer shell, and because it is a gas it moves very quickly which proves why we don’t see chlorine in its natural form floating around in nature. On the other hand, chemical property of chlorine is that it has immediate solubility when added to water. When chlorine is added to water, dissociation occurs and the water reacts to the chlorine. This reaction leaves us with HOCl (hypochlorous acid) + HCl (hydrochloric acid) (Becker, 2016). These are two very important compounds. The product of chlorine and water is actually more common than you would think, we can find them anywhere from bleach to swimming pools. Because when chlorine is added to swimming pools/water, hypochlorous and hydrochloric are created. Both products kill microorganisms and bacteria by attacking the lipids in the cell walls and destroying the enzymes and structures inside the cell (Unkown, 2014). Chlorine is very effective because chlorine (gas) is known to kill almost all kinds of bacteria (Becker, 2016). It is most effective when added at night. This is because it gets more time to act; in bright sunlight the hypochlorite ion breaks up very quickly (Unkown, 2014). Also, it leaves behind no contaminants in the water. Chlorine is more effective at night because in the daylight the heat would cause the bacteria to multiple. Although the heat would allow the atoms to move faster it wouldn’t be as fast as the rate of the multiplying of the bacteria. To connect chlorine’s effective to the particle model, we can see that chlorine gas atoms move very quickly and therefor can kill bacteria quicker than other disinfectants.