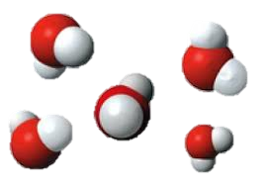
Particle Model
compoundelement |
Both elements and compounds are chemical substances found in the world. As seen in the particle model above, an element only includes the same atoms. In contrast, compounds are made up of different elements of which are chemically bonded together, to add to that point the diagram/model also shows that compounds always contain the same ratio of the atoms that make up the compound (Unknown, 2016). For example water always consist of a 2 hydrogen atoms and 1 oxygen ratio. As described in the diagram compounds consist of atoms of two or more different elements. These two different substance types can be distinguish differently as well. For example elements are distinguished by their atomic number whereas a compound is distinguished by the type of elements that makes up the compound (Unknown, 2016). An additional difference is that chemicals can be broken down into “simpler” substances such as an element by chemical changes/reactions and such, whereas elements cannot. This is because an element only consists of one type of atom (as illustrated in the particle model). We can see elements as atoms such as argon, or as molecules such as nitrogen gas (nitrogen gas includes two nitrogen atoms bonded together)
|
|
SALT-COMPOUND, CHLORINE-ELEMENT
Like the examples sated in the description of compounds and elements, sodium chloride and chlorine are yet another example. Sodium chloride is an compound, it consists of chlorine and sodium with the fixed ratio of 1:1. Salt is also a solid making the atoms inside of it very close together, denser and would have very slow movements (in the diagram to the left). Additionally the diagram also shows an equal of sodium and chlorine atoms. In contrast, chlorine which is an element that makes up salt, is a gas. In the particle model on the right, it shows chlorine gas atoms very far from each other and they would also move very quickly (Banks, 2016). Chlorine like any other element is identified by it's chemical name Cl, its atomic weight of 35 and number of 17. On the other hand the compound sodium chloride is identified by the specific atoms that are in it. Overall the particle model of the gas and element chlorine demonstrates the differences from the particle model of the compound sodium chloride. Differences include that the compound is a solid and it's atoms are much denser and have slow movements, whereas the elements atoms are spread apart and move quickly due to the fact that it is a gas (Banks, 2016). Additionally we can also see that the compound has a fixed ratio of sodium and chlorine. |
Salt compound particle modelchlorine element particle model |